|
One study evaluating over 300,000 pregnancies at one medical center for the incidence of AF showed that at baseline in 42.3 per 100,000 pregnancies, the women had a pre-existing diagnosis of AF before the pregnancy (112 total pregnancies in 93 women). The development of atrial fibrillation in women of childbearing age and during pregnancy is uncommon, although the incidence has been increasing over time. Atrial Fibrillation in Women of Child-Bearing Age & Pregnancy 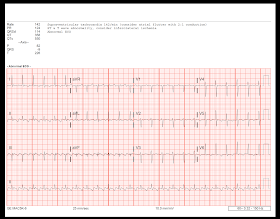
In the United States, the lifetime risk of AF is approximately 1 in 3 for White people and 1 in 5 for African American people, while increased socioeconomic status is associated with an increased risk of developing AF. However, its prevalence can vary based on gender, race, and a patient’s co-morbidities. Globally, age has the greatest contribution to the risk of the development of AF. For instance, in the United States, it is projected that the prevalence of AF will more than double between the years 20 (5.2 million in 2010 to an estimated 12.1 million in 2030). The incidence of AF was noted to be highest in the United States, Canada, Australia, and parts of Europe, while the lowest incidence was noted in Africa and the Middle East, with projections for this burden to increase in the future. Epidemiologic studies estimated the worldwide prevalence of AF was estimated to approximately 37.5 million in 2017, with approximately 3 million new cases every year. The increasing burden of AF has been a major concern. This update to the consensus guidelines for the management of AF has led to an increase in rhythm control in patients with AF, including the increased use of AADs alone or in conjunction with catheter ablations. This benefit of rhythm control has been most pronounced in certain populations, such as those with heart failure with reduced ejection fraction (HFrEF), leading to an update in our current clinical guidelines. However, as the efficacy of rhythm control strategies for AF has improved with the introduction of catheter ablation in the last two decades, such as the Early Treatment of Atrial Fibrillation for Stroke Prevention Trial, Atrial Fibrillation Network (EAST-AFNET) has shown a possible benefit of earlier more aggressive pursuance of rhythm control. In this context, providers often avoided the use of anti-arrhythmic drugs (AADs), which are often less well tolerated, require closer monitoring, and may be associated with several drug-to-drug interactions when compared to rate control agents. Initial trials such as Rate Control versus Electrical Cardioversion for Persistent Atrial Fibrillation Study (RACE) and Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM), have demonstrated rate control to be non-inferior to rhythm control. If the clinical momentum towards rhythm control continues, real-world data analysis will be needed to evaluate the clinical impact of the use, risk, and benefits of anti-arrhythmic medications. Providers must balance the increased emphasis on rhythm control with the overall clinical impact on their patients due to drug-to-drug interactions and adverse effects that can occur with different co-morbidities. As we enter the era of rhythm control, increased awareness is needed regarding the monitoring and potential adverse events that can occur with these medications. This paper will review the epidemiology and clinical impact of the utilization of anti-arrhythmic medications. 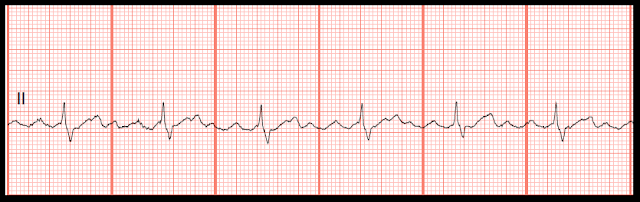
Because of this trend towards increased rhythm control, more patients have been placed on anti-arrhythmic medications. Initial trials have stated that rate control was not inferior to rhythm control, however, as the efficacy of rhythm control of atrial fibrillation has improved, a benefit in rhythm control has been shown. As the prevalence of atrial fibrillation continues to grow, multiple landmark trials have been designed to determine the best method to treat atrial fibrillation. Atrial fibrillation has been described as a global epidemic with a three-fold increase in prevalence in the last 50 years.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
